Chemical modification of PVC by different nucleophiles in solvent/non-solvent system at high temperature | SpringerLink

The solution containing 4.0gm of a polyvinyl chloride polymer in 1 litre dioxane was found to have an osmotic pressure 6.0xx10^(-4)atmosphere at 300K,the value of R used is 0.082litre atmosphere"mole"^(-1)K^(-1).The molecular mass

A solution containing 4.0 g of PVC in 2 litre of dioxane (industrial solvent ) was found to have an osmotic pressure 3.0xx10^(-4) atm at 27^(@)C The molecular mass of the polymer will be :

Comparison of glass transition temperature T g vs molecular mass M:... | Download Scientific Diagram

SOLVED: What is the degree of polymerization for polyvinyl chloride if its number average molecular weight is 16,537 g/mol?

SOLVED: Solutions of polyvinyl chloride (PVC) in cyclohexanone at 298 K were found t0 have the following solution heights for the given mass concentrations 9 L– hcm -00 0.30 2.00 0.75 3.00
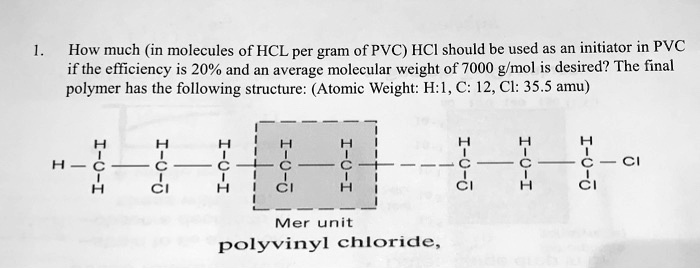
SOLVED: How much (in molecules of HCL per gram of PVC) HCI should be used as an initiator in PVC if the efficiency is 20% and an average molecular weight of 7000


![Solved Problem 5) Polyvinylchloride [PVC] is known to show a | Chegg.com Solved Problem 5) Polyvinylchloride [PVC] is known to show a | Chegg.com](https://media.cheggcdn.com/media%2Fd99%2Fd9995d25-f07e-40c0-819c-59c44fce7118%2FphpK1bBGn.png)