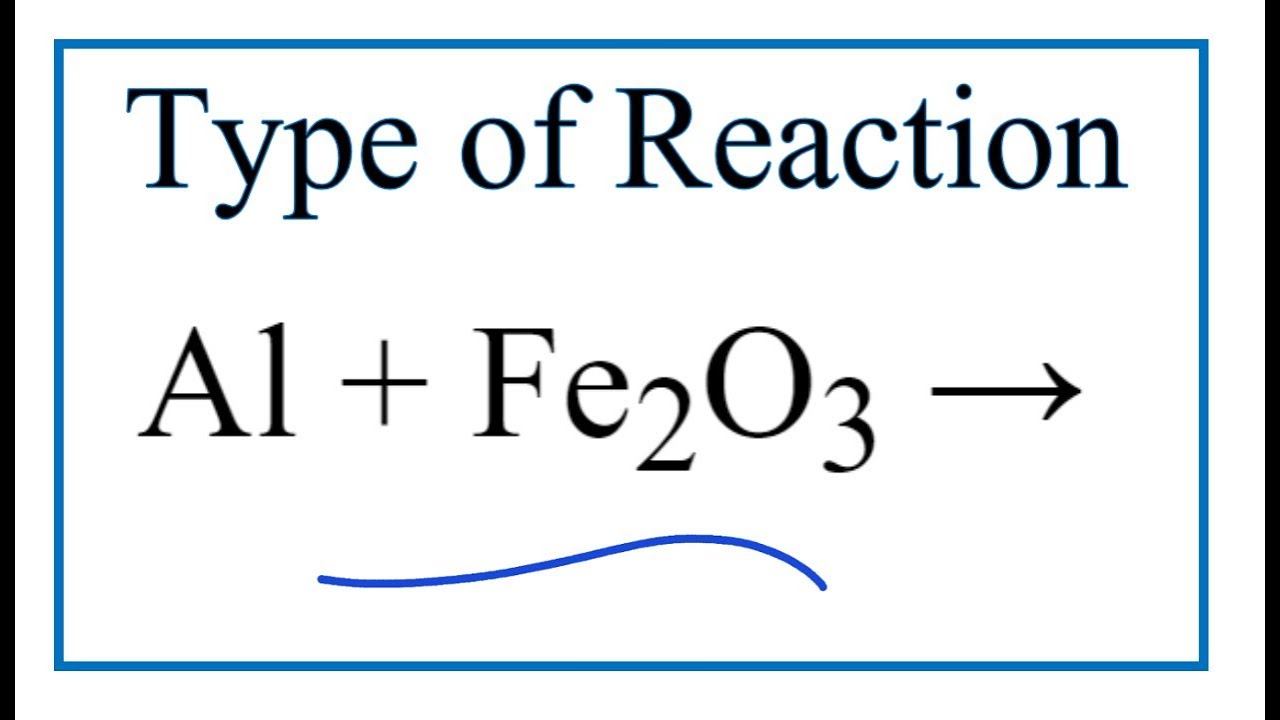An extensive study on the synthesis of iron based magnetic aluminium oxide nanocomposites by solution combustion method - ScienceDirect
SOLVED: Solid aluminum reacts with solid iron (III) oxide to produce liquid iron and aluminum oxide powder. Write a balanced chemical equation for this reaction showing the reactants and products. Include subscripts

If thermite is so easy to make, (powdered aluminum, iron oxide) and burns at thousands of degrees, why is it not used more in arson? - Quora
Displacement reactions of metal oxides - The reactivity series of metals - GCSE Chemistry (Single Science) Revision - WJEC - BBC Bitesize

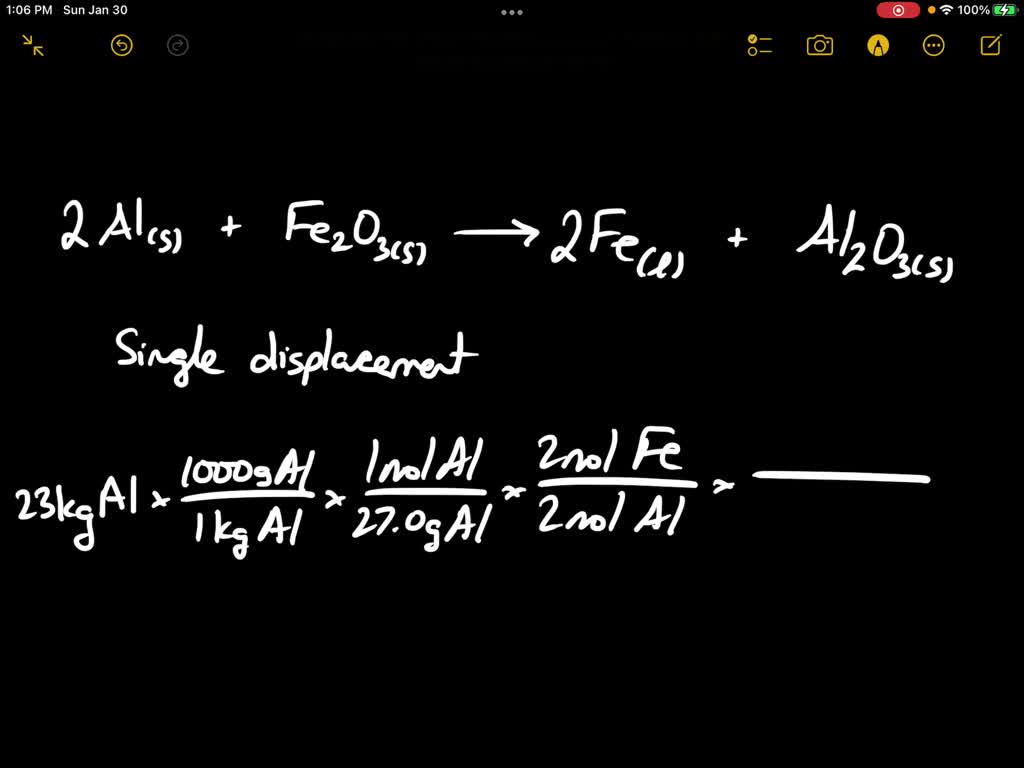
When a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and a...

SOLVED: The thermite reaction has solid aluminum powder (28.871 J/(mol·K)) reacting with iron(III) oxide (87.793 J/(mol·K)) to make aluminum oxide (49.56 J/(mol·K)) and iron (25.305 J/(mol·K)). The reaction is so exothermic that
![PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/035f93a77120a19b7cb355ec122296f5319649ce/3-Figure1-1.png)

:max_bytes(150000):strip_icc()/6247677404_ba1fe32d71_k-9925d5bcbd0a4668bab07102590c6a81.jpg)