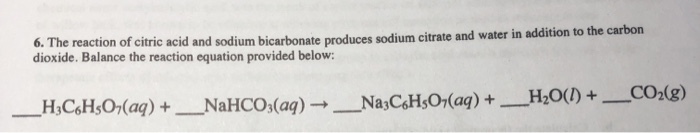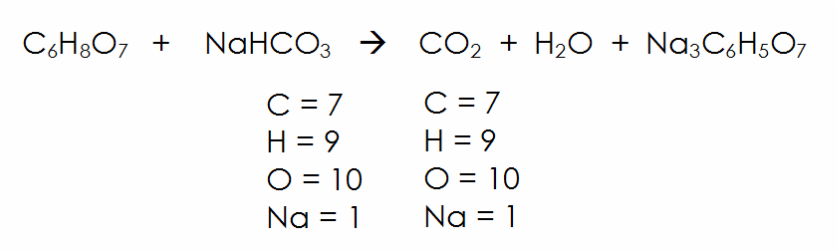A pneumatic power source using a sodium bicarbonate and citric acid reaction with pressure booster for use in mobile devices | Semantic Scholar
Is the reaction bicarbonate with citric acid exothermic or endothermic? My teacher and my text book were saying it is exothermic, but I got results showing it is endothermic. - Quora

A pneumatic power source using a sodium bicarbonate and citric acid reaction with pressure booster for use in mobile devices | Semantic Scholar