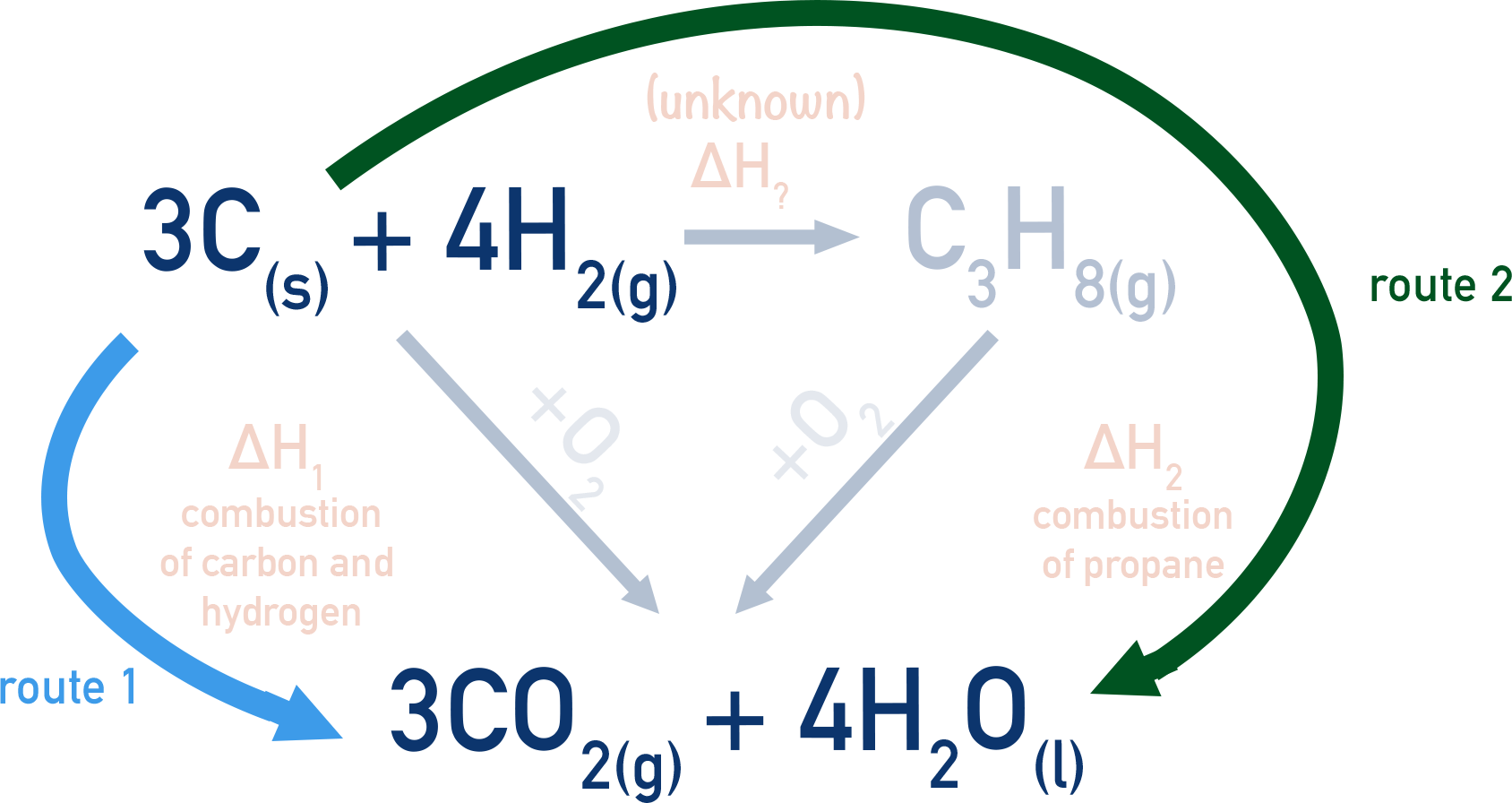
JUHTUVUI Volume (V) Calculate the enthalpy of formation of Benzene (C Hz), standard enthalpies of combustion of Carbon (C), Hydrogen (Hz) and benzene (CH) are -393.5 Kj mol-1 - 285.83 Kj mol-

The enthalpies of formation of carbon nanomaterials as a key factor for understanding their structural features - Physical Chemistry Chemical Physics (RSC Publishing)
CALCULATE THE HEAT OF FORMATION OF CARBON MONOXIDE FROM THE FOLLOWING DATA 1.C+O2->CO2 dH=-93.5kj 2.CO+1/2O2->CO2 dH=2.8kj - 7splwv99
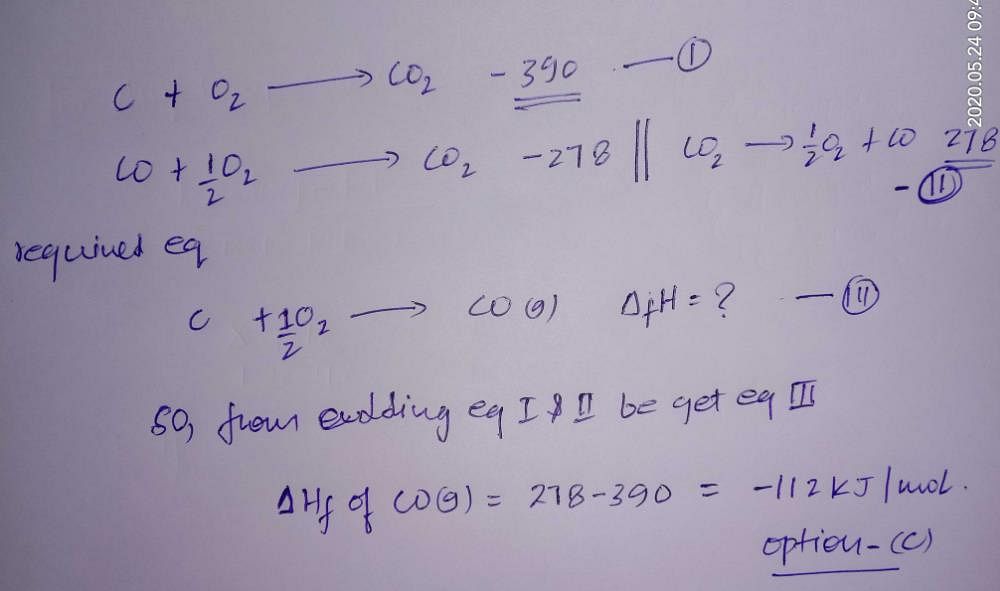
The enthalpies of combustion of carbon and carbon monoxide are -390 kJ mol1 and -278 kJ mol1 respectively. The enthalpy of formation of carbon monoxide isa)668 kJ mol1b)112 kJ mol1c)-112 kJ mol1d)-668
66 Enthalpies of formation of co(g),CO2(g),N2O(g)and N2O4(g) are 110, 398, 81, 97 KJ/lol respectively. Find value of H for the reaction. N2O4(g) +3CO(g) — >N20(g)+3CO2(g)

The heat of the combustion of graphite and carbon monoxide respectively are 393.5 kJ mol1 and 283 kJ mol ^{ -1 }. Thus heat of formation of carbon monoxide in kJ mol ^{ -1 } is:-110.5+110.5-55.25None of these
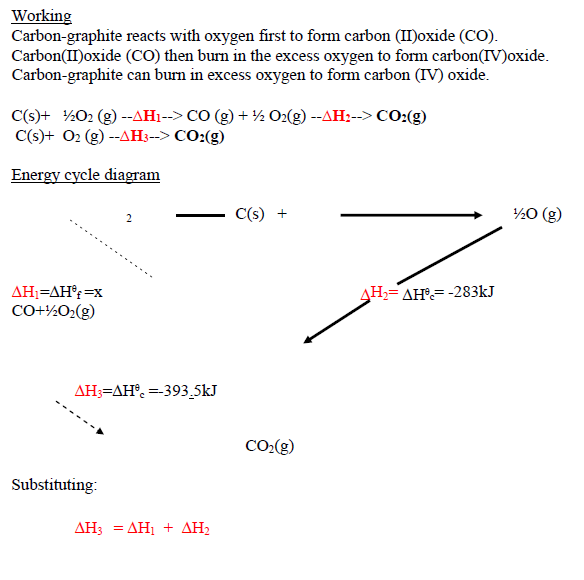
Calculate the molar enthalpy of formation of carbon(II)oxide (CO) given : ?H?c of carbon-graphite = -393.5kJmole<sup>-1</sup>, ?H?c of carbon(II)oxide (CO)= -283 kJmole<sup>-1</sup>
![PDF] Prediction of Enthalpy of Formation in the Solid State (at 298.15 K) using Second-Order Group Contributions. Part 1. Carbon-Hydrogen and Carbon-Hydrogen-Oxygen Compounds | Semantic Scholar PDF] Prediction of Enthalpy of Formation in the Solid State (at 298.15 K) using Second-Order Group Contributions. Part 1. Carbon-Hydrogen and Carbon-Hydrogen-Oxygen Compounds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c766ff3889423f02f154fa43603214c3fec1f15d/4-Table1-1.png)
PDF] Prediction of Enthalpy of Formation in the Solid State (at 298.15 K) using Second-Order Group Contributions. Part 1. Carbon-Hydrogen and Carbon-Hydrogen-Oxygen Compounds | Semantic Scholar