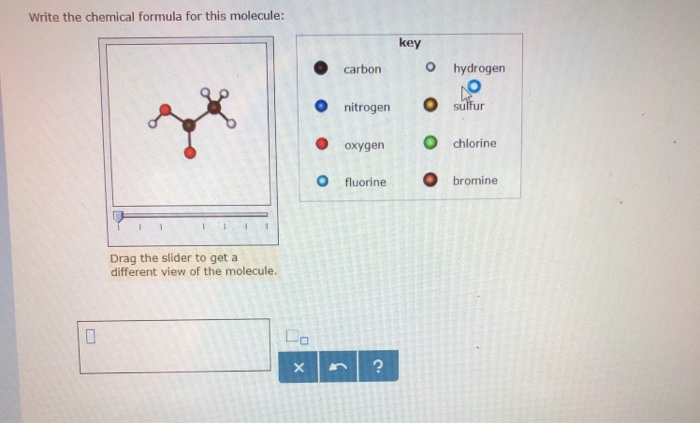
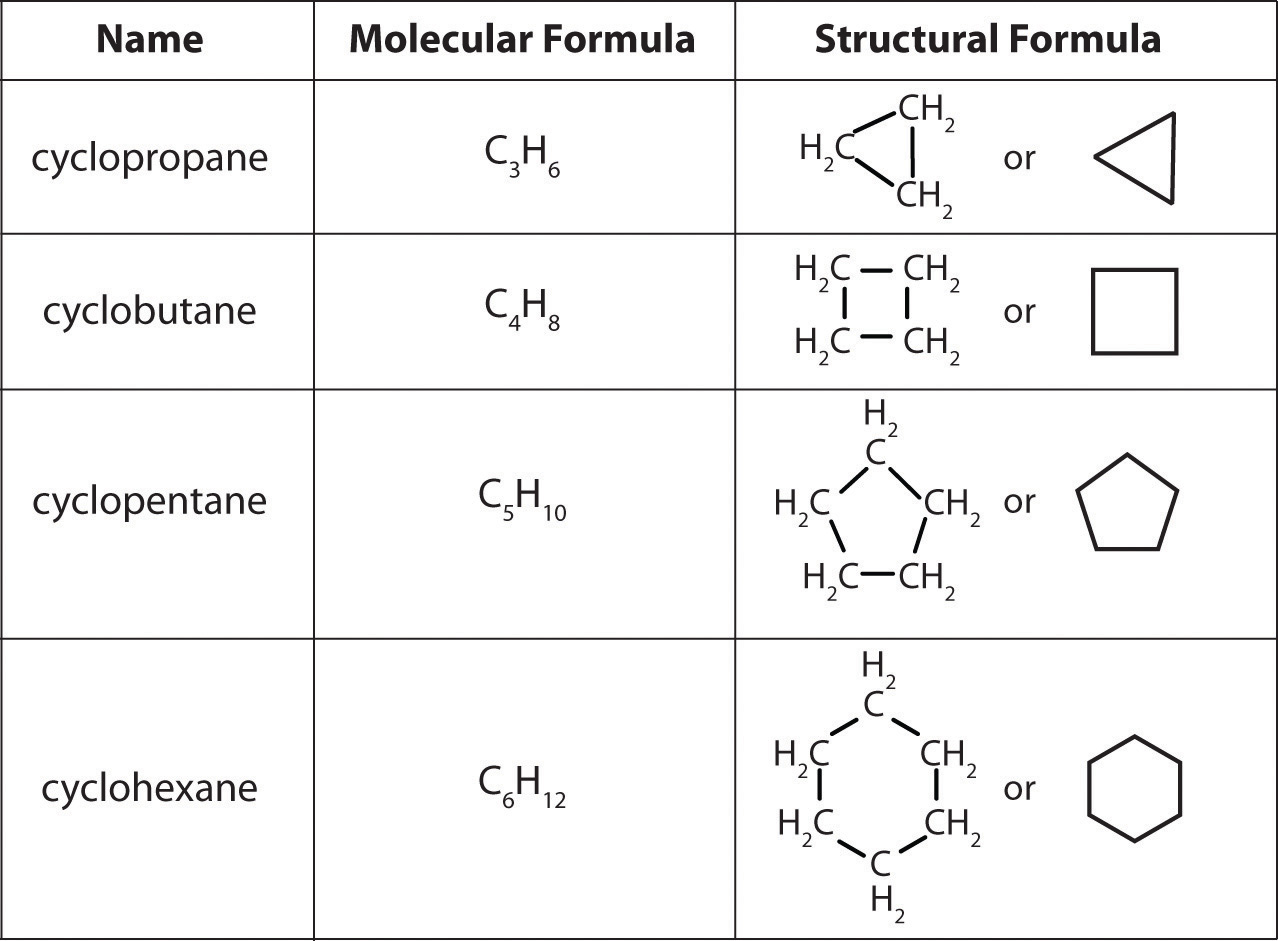
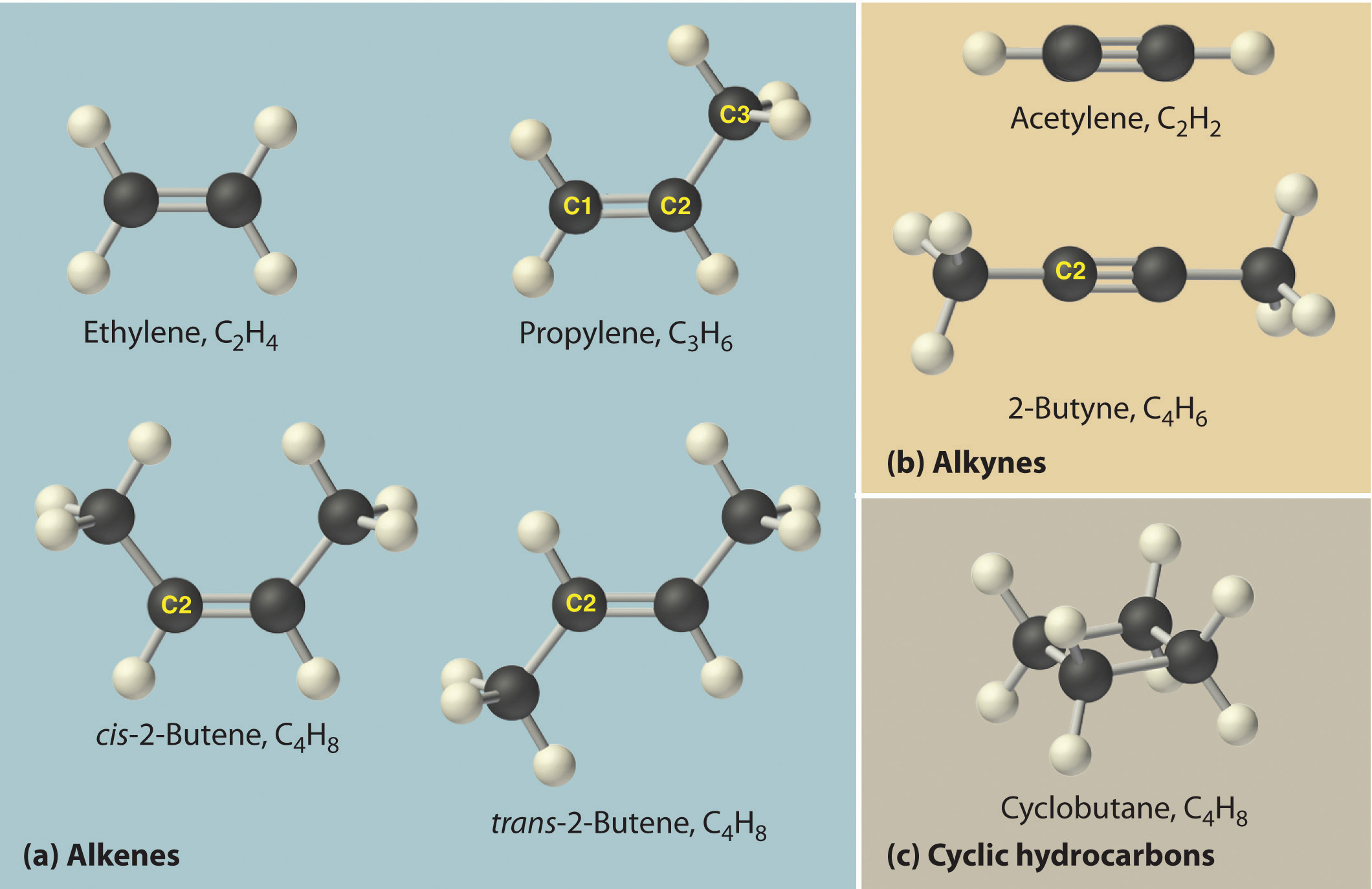
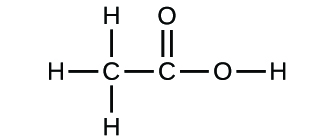
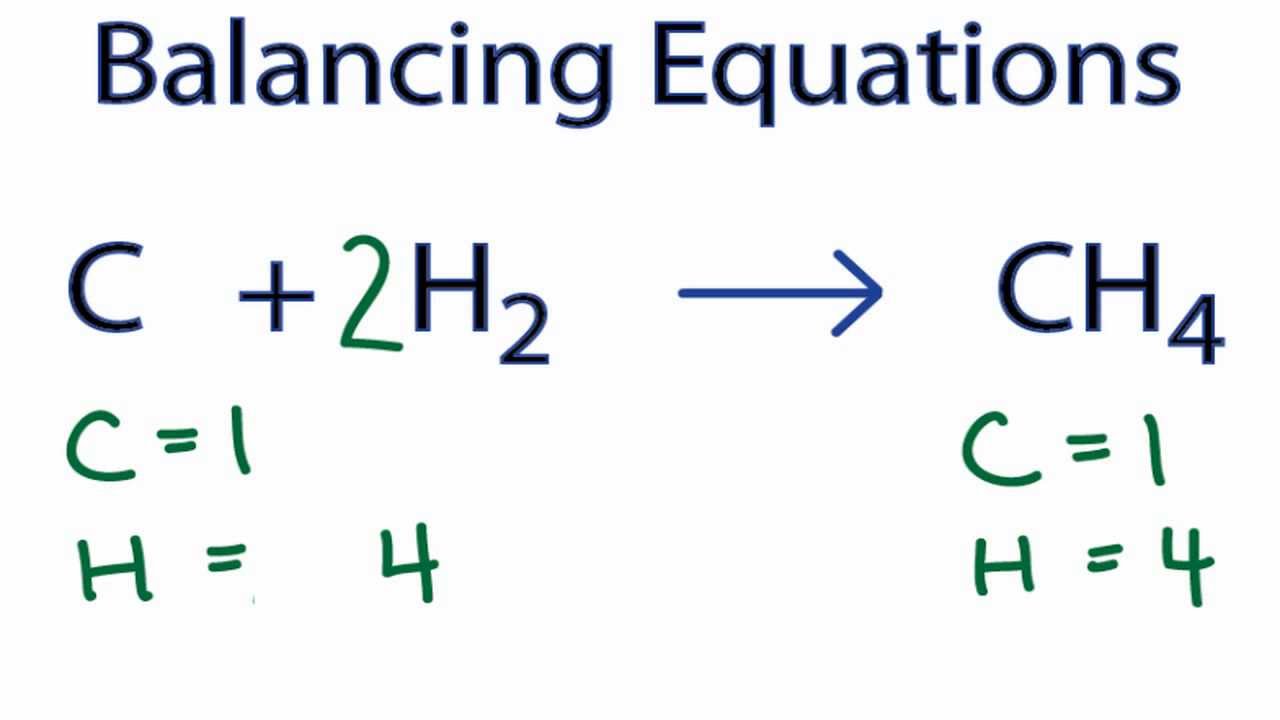The empirical formula of an organic compound containing carbon and hydrogen is CH2 . The mass of one litre of this organic gas is exactly equal to that of one litre of

Question Video: Determining the Number of Carbon Atoms in an Alkene Given the Number of Hydrogen Atoms | Nagwa

An organic compound contains 20 % carbon, 6.7 % hydrogen, and 46.67 % nitrogen. Its molecular weight was found to be 60 . Find the molecular formula of the compound.

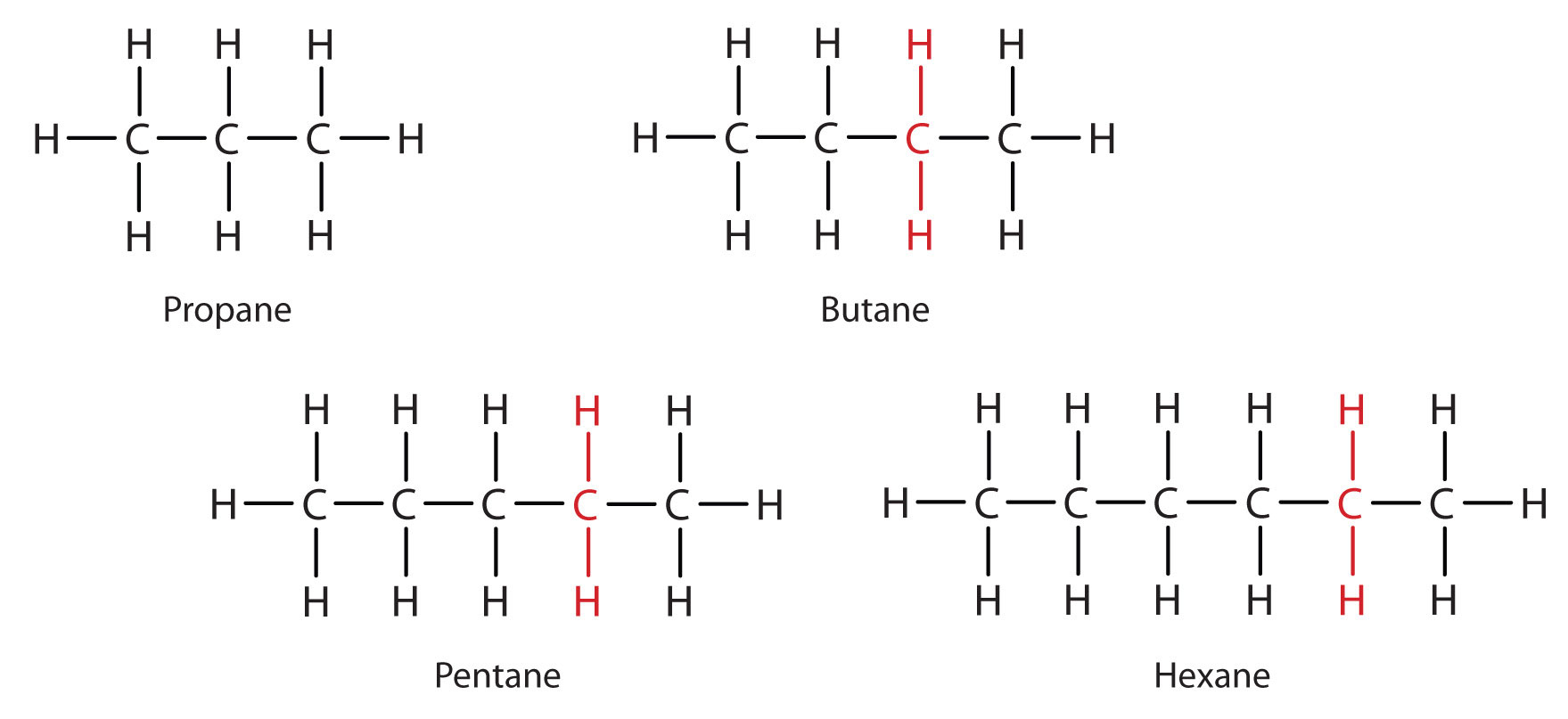
How many hydrogen atoms are present around each highlighted carbon atom in the following molecules? What is the molecular formula for each molecule? Both compounds are active ingredients in some common sunscreens.

Calculate the Empirical Formula for a compound with the following composition: 46.16% carbon; 53.84% nitrogen 1)Change % to grams (if needed) 2)Convert. - ppt download

An organic compound contains carbon, hydrogen and oxygen. Its elemental analysis gave`C,38.71% - YouTube

Table of atoms in molecules, chemical formula of carbon,oxygen,hydrogen and nitrogenmolecules.Educational and study content of chemistry and science s Stock Vector Image & Art - Alamy