Atomic masses L.O.: Define the terms relative isotopic mass and relative atomic mass, based on the 12C scale; Calculate the relative atomic mass of. - ppt download
The table below show the relative atomic masses and the percentage abundance of the isotopes L1 and - Tutorke

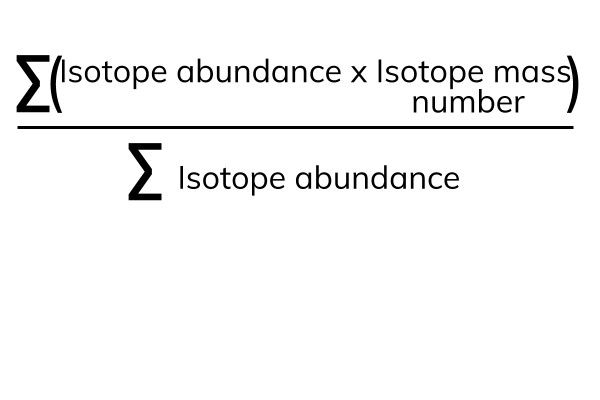
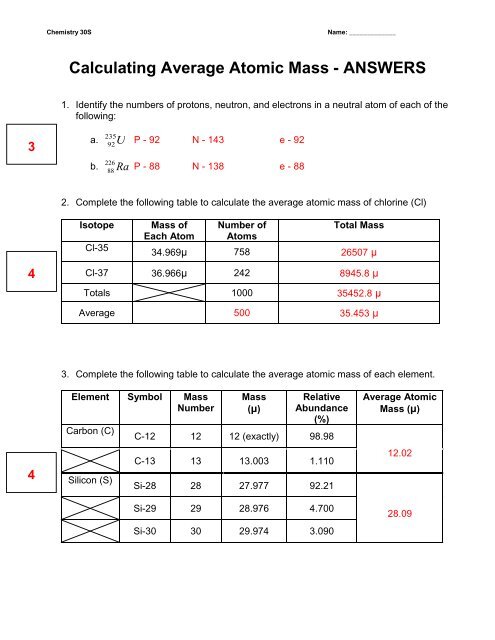
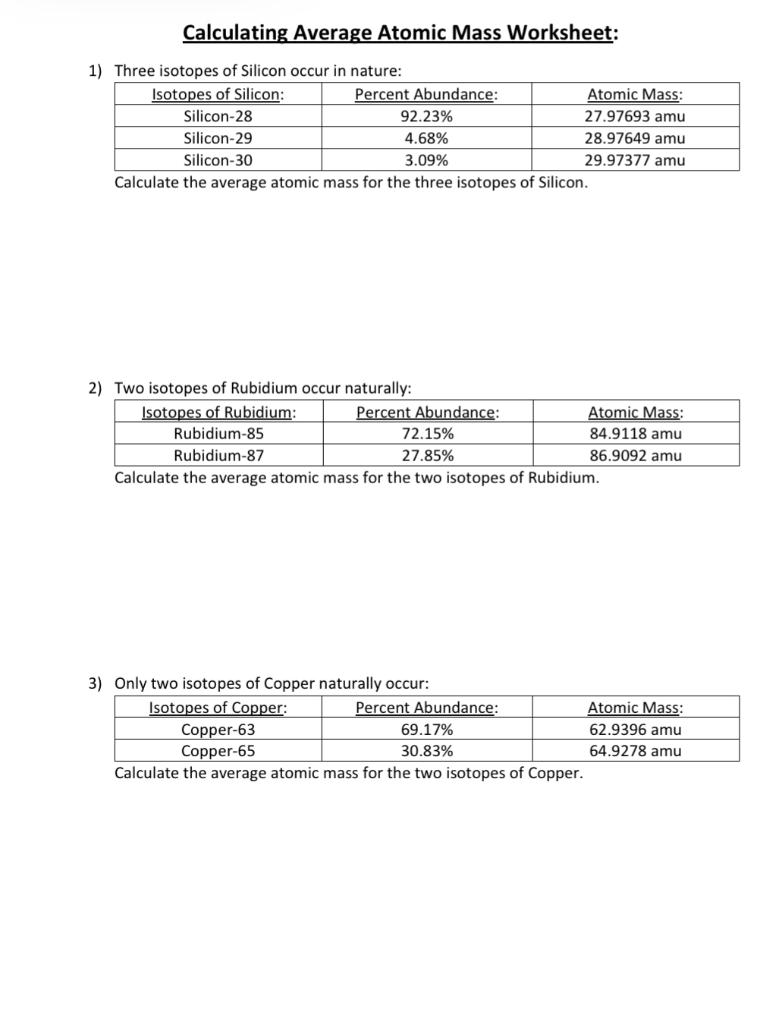
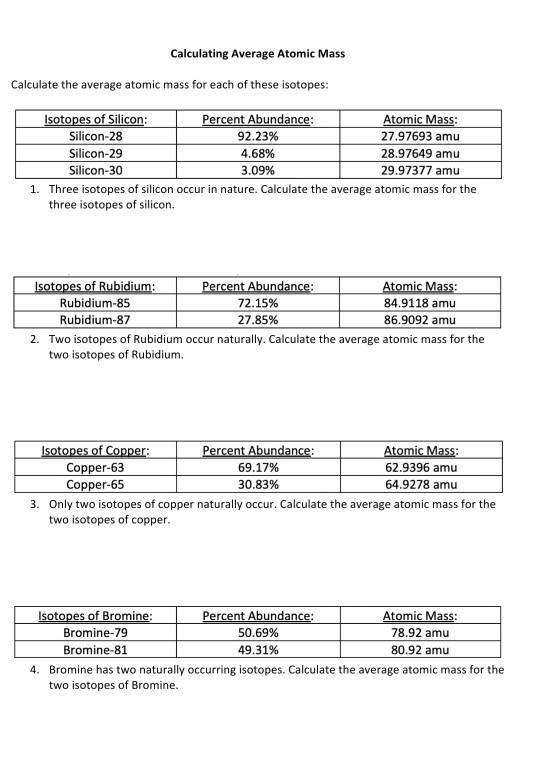
Average Atomic Mass In nature, most elements are a mixture of different isotopes The mass of a sample of an element is a weighted average of all the isotopes. - ppt download
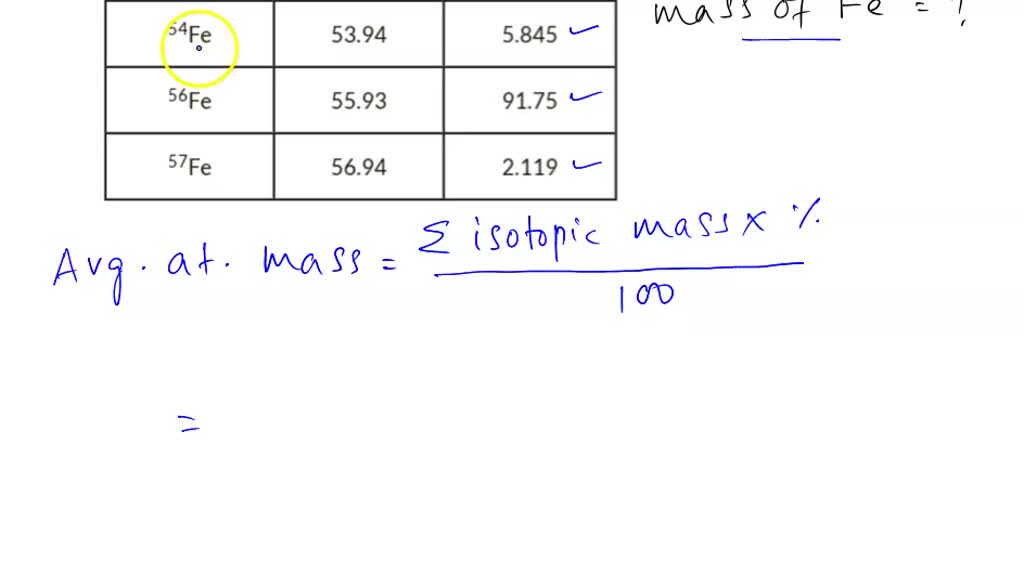
SOLVED: Calculate the average atomic mass of iron using the isotopes in Table 2 below: Table 2. Stable Isotopes of Iron Isotope Mass (amu) % Abundance 54Fe 53.94 5.845 56Fe 55.93 91.75 57Fe 56.94 2.119

Isotopic Abundance & Relative Atomic Mass (4.1.3) | CIE AS Chemistry Revision Notes 2022 | Save My Exams
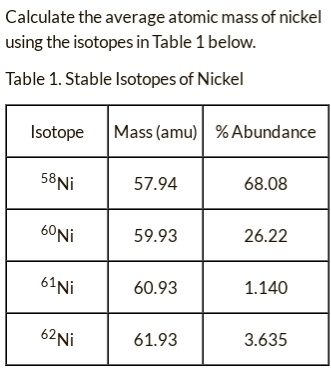
SOLVED: Calculate the average atomic mass of nickel using the isotopes in Table 1 below: Table 1. Stable Isotopes of Nickel Isotope Mass (amu) % Abundance 58Ni 57.94 68.08 60Ni 59.93 26.22 60.93 1.140 62Ni 61.93 3.635 61Ni