Coatings | Free Full-Text | Evaluating the Protective Effects of Calcium Carbonate Coating on Sandstone Cultural Heritage

Difference Between Calcium Chloride and Potassium Chloride | Compare the Difference Between Similar Terms

SEM image showing the effects of the use of different calcium salts on... | Download Scientific Diagram
Q. Calcium carbonate reacts with aqueous HCL to give Calcium chloride and carbon dioxide ,according to the reaction CaCo3(s)+2HCl(aq) →CaCl2(aq)+CO2(g)+H2O(l) What mass of calcium carbonate is required to react completely with 25

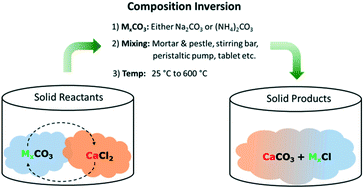
Composition inversion to form calcium carbonate mixtures - CrystEngComm (RSC Publishing) DOI:10.1039/C7CE00433H
48 CaCO + 2HCl >CaCl + CO+ HO ; In this chemical reaction what will be the amount of calcium chloride when 10g calcium carbonate and 400 mL 0.25 M HCl is used : (1) 4.05 g (2) 5.55 g (3) 11.1 g (4) 16.65 g

Difference Between Calcium Chloride and Potassium Chloride | Compare the Difference Between Similar Terms

Forests | Free Full-Text | Hot Compression of Calcium Chloride and Sodium Carbonate Modifies Wood for Tsoongiodendron odorum

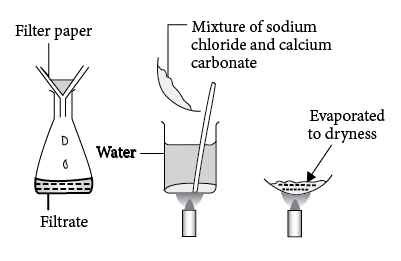
Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution