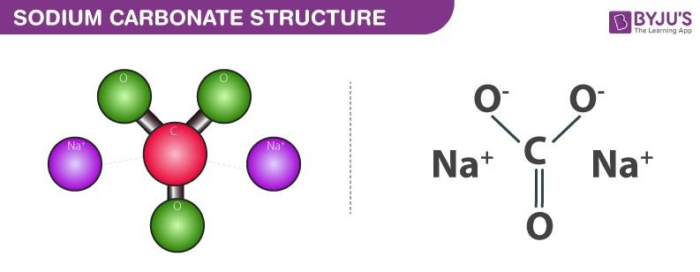
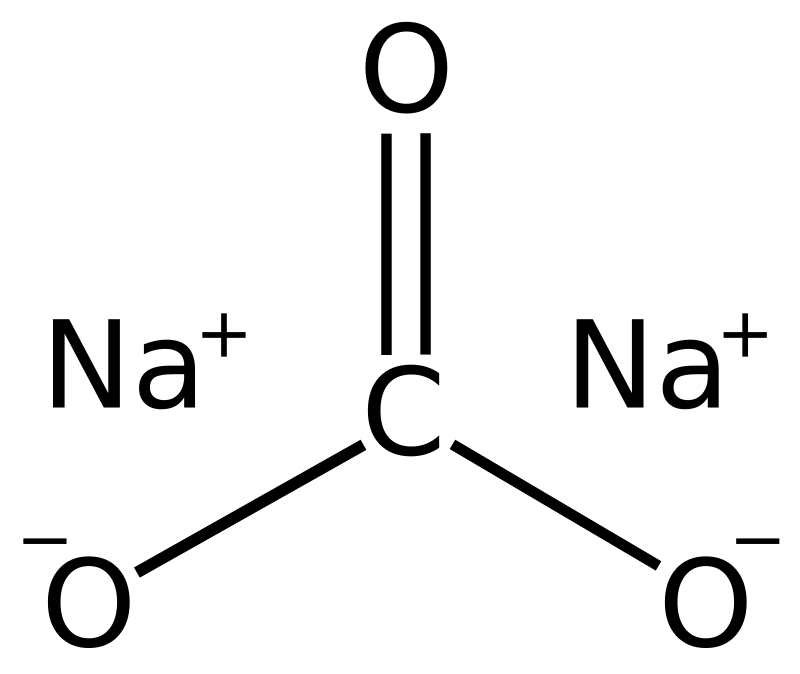
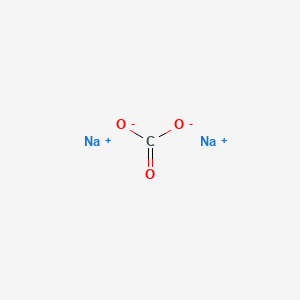
Please solve 40,41,42 37 38 39 40 41 42 43 Determine the percentage - Chemistry - Some Basic Concepts of Chemistry - 12637943 | Meritnation.com

Speci…c heat capacity for the anhydrous sodium carbonate. The inset... | Download Scientific Diagram

What is the formula mass of anhydrous sodium carbonate ? (Given that the atomic masses of sodium... - YouTube
SOLVED: 12 g of a mixture of anhydrous sodium carbonate (Na2CO3) and sodium chloride (NaCl) (as impurity) were made up to 1 dm^3 of aqueous solution. 25 cm^3 of this mixture was

Please solve 40,41,42 37 38 39 40 41 42 43 Determine the percentage - Chemistry - Some Basic Concepts of Chemistry - 12637943 | Meritnation.com

25 mL of a mixed solution of sodium carbonate and sodium bicarbonate required 10 mL of N/20 HCl when titrated in the presence of phenolphthalein but 25 mL of the same when
Determine tge percentage composition of a mixture of anhydrous sodium carbonate and sodium bicarbonate from the following data : weight of mixture taken = 2g loss in weight on heating = 0.11

2.650 g of anhydrous sodium carbonate are dissolved in water and the solution made up to 500 mL. On titration, 50 mL of this solution neutralised 50 mL of a solution of
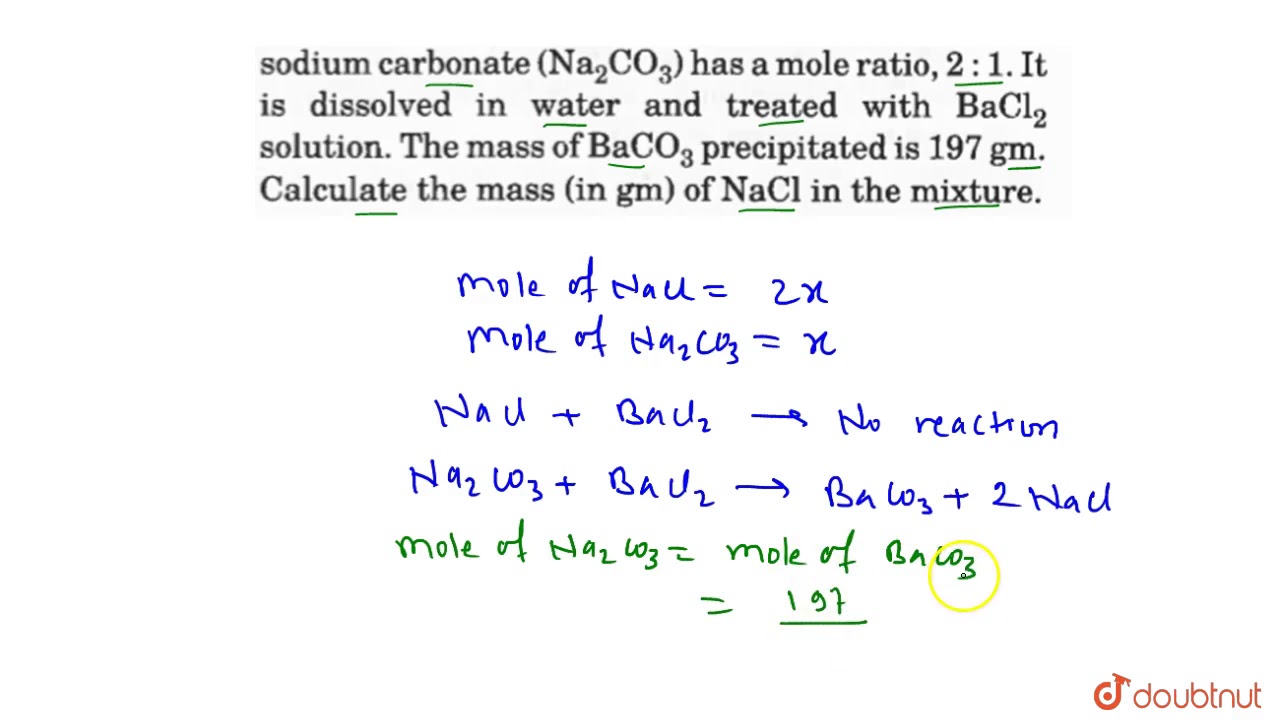
A mixture of sodium chloride(NaCl) and anhydrous sodium carbonate `(Na_(2)CO_(3))` has a mole ratio, - YouTube

A mixture of sodium chloride (NaCI) and anhydrous sodium carbonate (Na,cO,) has a mole ratio 2:1. It is dissolved in water and treated with BaCl2 solution. The mass of BaCO, precipitated is
![chemistry - stoichiometry] im very stuck pls help : r/HomeworkHelp chemistry - stoichiometry] im very stuck pls help : r/HomeworkHelp](https://preview.redd.it/j3h6rg0fx5n81.jpg?width=640&crop=smart&auto=webp&s=03416edb596c63219237b91215197437fa74eef4)